By Savannah Gleim & Stuart Smyth
What came first, science or regulation?
When you think about safety, you might think that regulation should lead science. Yet, it frequently happens the other way around. Science often outpaces regulation, resulting in rules and assessments needing to be developed to ensure safety. When science and innovation push beyond existing regulations, existing frameworks must either be adapted to fit the new innovation or be scrapped and newly built.[1].
Biotech Underpinning
In examining the regulatory framework of genetically modified (GM) crops, we asked what underpinning science was used to first develop regulations. Discovering the underpinnings of initial regulations may help to provide unique insights into how science forms the basis for policy development within innovative sectors such as GM crops and can be used as evidence to reassure consumers. This led to our research “Scientific underpinnings of biotechnology regulatory frameworks”, which has been published in New Biotechnology.
In 1992, the Flavr Savr tomato was approved in the USA (no longer being produced), marking it as the first biotech approved crop in the world. Two years later, GM canola was approved in Canada, which is still grown today. Leading up to these production approvals, regulations needed to be created or modified to determine the safety of biotech innovations. In search of regulatory documents, we looked at the early adopters, Canada, European Union and the USA, as well as the international organizations that provided guidance documents, such as the Organisation for Economic Co-operation and Development , searching not only the regulations, but also evidence of scientific research from conferences/workshops and publications.
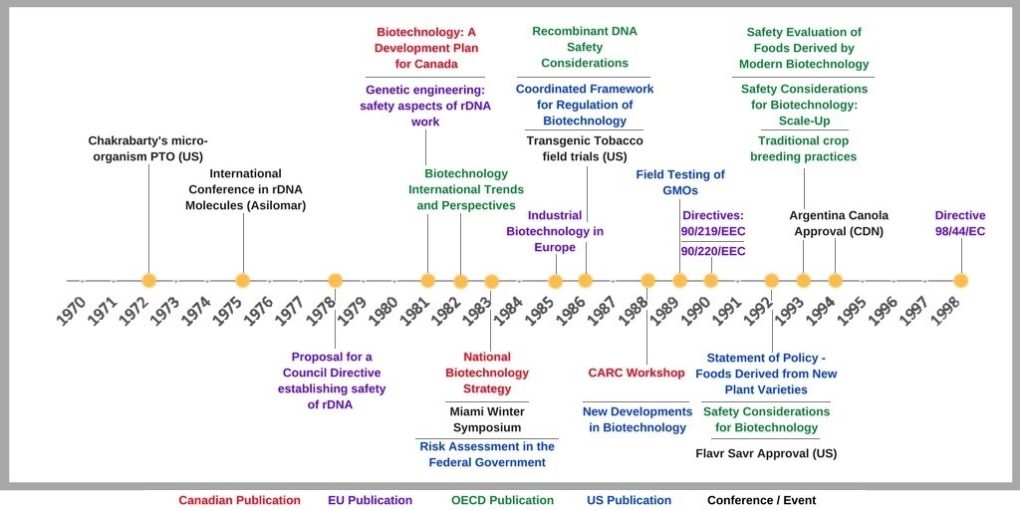
Even though we work at a university, which has many resources and documentation, finding evidence was not easy. Prior to the Internet, scientific evidence used for regulation was based on what was published in journals, presented at conferences and workshops, or from relationships regulators had with the predominantly local scientific community. Below is a timeline of events, regulations, and publications we were able to find.
Transparency blunder of policy
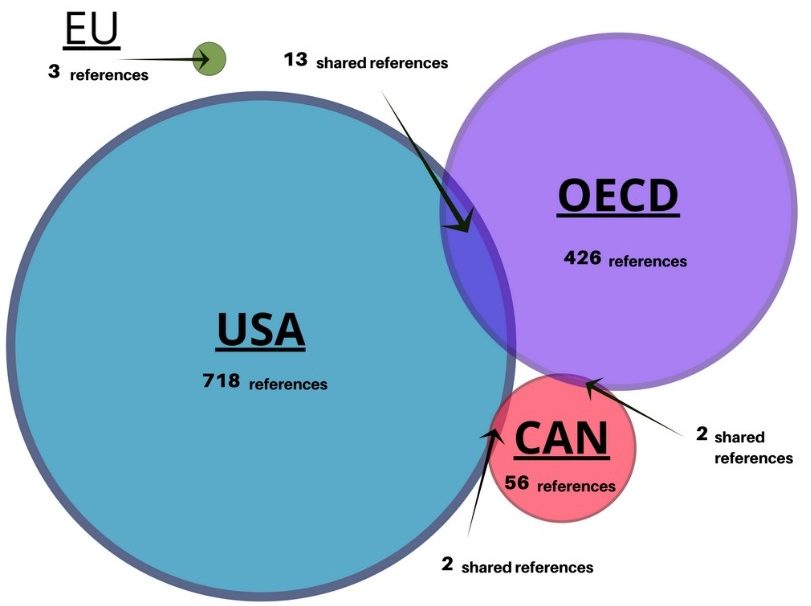
We invite you to read our paper and other results, as we aren’t able to report everything in one blog. What became apparent in our research is that legislation is not a good source of information, it’s the nature of how legislation is written, there are no references. For example, the format of the Canadian Seed Act of 1985 doesn’t account for any references or scientific evidence to support the Act, it’s merely a legal document. That’s what led us to expand our research beyond Acts and regulations to include publications, conferences and workshops involving and often organized by regulators, to gain evidence about the science, which directed regulation development. From the 16 documents, we found a significant number of scientific references 989 references to publications, 718 alone within the USA publications. Yet, there was little overlap of scientific underpinnings used between the four jurisdictions as shown in the Venn diagram.
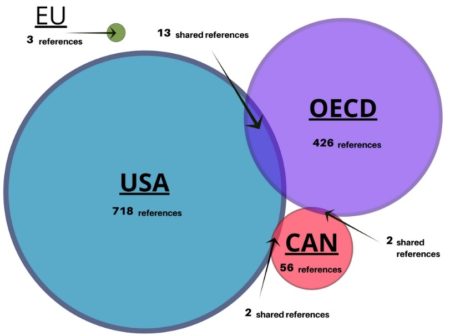
We found that at the time of regulatory framework development, scientific research underpinned regulation. Scientific evidence was used to identify what potential risks could exist and how to best assess them. It also seems that the relationship between regulators and public and private sector scientists played an important role the development process. In numerous reports, government officials and scientists participated and offered expertise. Regulation of biotechnology is underpinned by knowledge exchange and information sharing between regulators and public and private sector experts.
Our research continues
The search for scientific underpinnings of biotechnology regulation does not stop here. We are continuing to search for further documentation and evidence of science that has shaped regulation. If you have any documents or suggestions of policy, acts, directives, conference/workshops/symposiums, please feel free to share them with us, as we wish to better understand not only what science has shaped regulation, but how biotechnology research was shared between countries and regulatory agencies.
Reference:
Gleim, S., & Smyth, S. J. (2018). Scientific underpinnings of biotechnology regulatory frameworks. New Biotechnology, 42, 26–32. https://doi.org/10.1016/j.nbt.2018.01.004
[1] The European Union is an example of creating a new regulation system for the risk assessment of genetically modified crops.
[2] See Table 2 for the full list of shared or Cross-References Publications underpinning regulations amongst Canada, USA, OECD and the EU.